Since German Nobel laureate Paul Ehrlich developed the concept of the “magic bullet” in 1907, researchers have chased the possibility relentlessly. Ehrlich’s immune response research proposed killing specific microbes without harming the body itself, a goal which...
Jesper Wrangle
6 Essential Qualities in a Drug Safety Assessment Testing Partner
Drug safety assessment is a highly complex, involved and molecule-specific process, which is why most drug developers benefit from working with a testing partner. But not all CROs are created equal. Here are six characteristics to consider. Safety assessment studies –...
What Is Bioanalytical Testing & Method Validation?
To be effective and safe, a therapy must contain precise drug concentration. That’s why bioanalytical testing is required during the drug development process. For robust and accurate data, bioanalytical testing relies on effective method development and validation. In...
QWBA vs. Mass Balance Studies
Quantitative whole-body autoradiography (QWBA) and mass balance testing are equally important processes to gather data on a drug candidate’s absorption, distribution, metabolism, and excretion (ADME) properties. While both QWBA and mass balance can give details...
What Is IND-Enabling Testing & What Does It Include?
IND-enabling testing is a cornerstone of the drug development process, helping researchers predict safety concerns and estimate safe and efficacious starting doses for clinical trials. Planning ahead, understanding the tests you need to conduct, and diligent tracking...
In Vivo ADME Testing in Drug Development: A Short Guide
In vitro and in vivo ADME studies provide valuable data about a test article as it moves through preclinical development. In this short guide, we provide an overview of in vivo ADME testing, including why it’s important and how it works. For a successful...
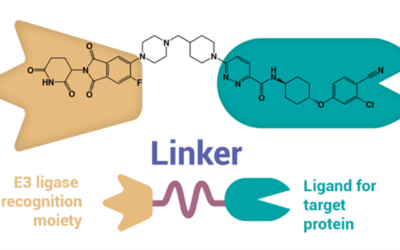
How to Improve PROTACs’ Oral Bioavailability
PROteolysis TArgeting Chimera (PROTAC)* drugs hold enormous potential. No PROTAC drugs have earned FDA approval yet, but they are being widely explored across industry and academia for an ever-expanding range of treatments. Part of PROTACs’ appeal is in their...
How to Tackle the Developmental Challenges of PROTAC Drugs
PROteolysis TArgeting Chimera, the novel therapeutic modalities better known as PROTACs*, first emerged in 2001 but are just starting to emerge from clinical trials. Their unique structure and capabilities have excited drug developers; however, there are still...
A Guide to In Vitro ADME Testing in Drug Development
Understanding the Absorption, Distribution, Metabolism and Excretion (ADME) properties of a compound is critical to drug discovery and development. In this short guide, we look at how in vitro ADME testing works and why it is important. To understand in vitro ADME...
Maximizing the Efficiency of Ocular Gene Therapies
Gene therapy is an exciting—and relatively new—area of medicine. Treatments were first introduced in the 1970s, but after decades of methodology challenges and scientific frustration, the concept of altering genes to address “untreatable” viruses and diseases gained...
Preparing Drug Developers for Developmental and Reproductive (DART) Studies
In 1993, the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) published guidance entitled “S5 Detection of Toxicity to Reproduction for Medicinal Products.” The U.S. FDA interpreted the ICH guidance and...
WuXi AppTec Expands Toxicology Footprint, Capabilities with New Chengdu Facility
A new 108,000-square-foot testing facility in Chengdu, China, will allow WuXi AppTec to help meet high global demand for toxicology testing in preclinical studies. The facility significantly expands WuXi AppTec’s testing capacity at a time when many customers’...