Extractables and Leachables Testing
Comprehensive E/L Studies
Accurate risk assessment requires full chemical characterization and a comprehensive approach to extractables and leachables testing. At WuXi AppTec, our goal is to develop an extractables/leachables (E/L) program that will pass muster with the evolving scientific and regulatory landscape. We have the team to ensure your test article is handled with the endgame in mind, from extraction through analytical testing to data interpretation. Extractables/leachables testing is about finding the chemicals you did not expect to find. That’s why we built a lab and E/L program based on the idea that complete identification should be the standard expectation – not an “add on” service.
- Single-use systems
- Container closure
- Delivery systems/Combination products

A Global Leader in Complete Identification

WuXi AppTec is the industry leader in E/L testing and unknown compound identification. Our state-of-the-art Chemical Characterization Center of Excellence, located in Saint Paul, MN, is an FDA-registered testing facility entirely dedicated to E/L studies. Out of this lab, our expert team performs all E/L testing associated with single-use systems (SUS) used in biopharma manufacturing, container closure along with stability testing, and combination products/delivery systems.
E/L Timeline of Guidance & Regulatory Standards
Extractables/leachables guidance and regulatory standards continue to evolve. We’ll work closely with your team to help interpret this guidance in a way that keeps your project moving forward.
Our E/L Experience
6,000+ E/L Programs
- Single-Use Systems
- Drug Delivery Systems
- Container Closure
- Combination Products
- Medical Devices
Complete Characterization
- Full chemical characterization and compound identification
- 20+ experts in data analysis and elucidation
- Commitment to no unknowns
1,200+ Safety/Tox Risk Assessments
- Experienced toxicology team
- Dedicated to writing safety/toxicology risk assessments
E/L Programs
Square feet of dedicated E/L testing space
Safety/toxicological risk assessments
Program Capabilities
From Single Use Systems to container closure and delivery systems, we can help you develop a comprehensive E/L program that keeps pace with the continuously changing scientific and regulatory landscape and also meets the needs of your specific project timeline and requirements.
E/L for Single Use Systems
The components in a biopharmaceutical manufacturing process are increasingly constructed of plastic materials. These components may contact multiple process streams, from raw material to final product. As such, determining the impact that extracted compounds from these plastic components (i.e., additives, oxidizers, plasticizers, etc.) have on the process streams aids in thoroughly assessing risk for the components in a system.
The risk-based approach will determine what testing is required for which system components. Not all plastic is created equal, so manufacturers must consider the plastic type, contact duration, temperature and proximity to the final drug to assess risk accurately. Conducting analytical evaluations for extractables and leachables for every component could be prohibitively expensive and highly time-consuming. However, certain products are considered high-risk and may warrant E/L analysis in greater depth (e.g., components with high surface area). Conversely, material with a low surface area and short contact time may be lower risk and require less analysis.
The components of a SUS are categorized into several types—e.g., bags, tubes, filters, connectors, valves, etc.—each of which requires a specific testing strategy. USP chapter <665> and the BPOG protocol provide in-depth guidance on time points, extraction solvents and testing protocols for each type of plastic component.
E/L for Container Closures and Combination Products
Similar considerations must be made for container closure and combination products. Working in collaboration with the manufacturer, the testing lab should determine the best route forward for extractables testing depending on the information available on the component’s materials of construction. Extractables profiling is highly complex, and the analytical techniques, solvents, time and temperature to be chosen will depend on the test article and the expected extractables from the information provided by the manufacturer.
Leachables testing on these products becomes even more complex due to the possibility that actual drug products will be used for the testing. Leachable compounds do not only carry with them their own toxicological impact but could also influence the efficacy of the drug product.
Our E/L Process
You cannot accurately assess safety without knowing the chemical constituents in your test article. Our experts harness mass accurate data, extremely sensitive and finely tuned equipment, a sizable proprietary LC-MS database and library of confirmed compounds, and expert compound elucidation to identify the chemicals at or above the analytical evaluation threshold (AET) for your test article. All within a program tailored to your unique product and program needs.
WuXi AppTec has a long-established reputation for a commitment to complete identification – to our customers and regulatory bodies. We find ourselves regularly helping customers with their regulatory challenges as a result of work from labs that are not committed to complete characterization.
We have developed a proprietary LC-MS database of more than 6,000 compounds. And we maintain a LC-MS library of more than 700 compounds verified through reference standards, as well as a team of expert scientists who can elucidate compounds not listed in either database. This makes WuXi AppTec’s LC-MS capability significantly more efficient and accurate than our competitors.

Understand Materials

Develop Mass Accurate Data

Search & Match Through Databases

Search Public Literature

Compound Elucidation
Regulatory Support & Guidance
WuXi AppTec provides regulatory support throughout the testing and regulatory review process to answer questions or supply justifications along the way. Our experts stay up to date on all current E/L trends and guidelines and work with regulatory agencies to ensure we can design programs that help you anticipate and meet specific needs.
Comprehensive E/L Studies
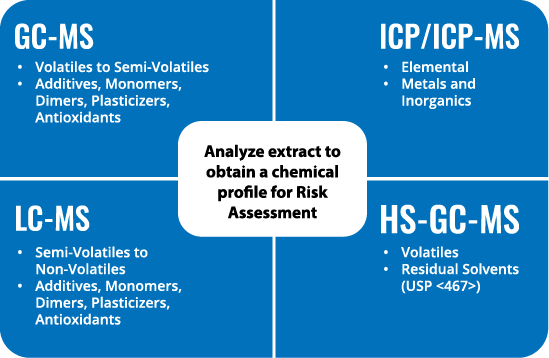
Analytical Approach
We offer a robust analytical approach that includes GC-MS, ICP/ICP-MS, LC-MS, and HS-GC-MS.
State of the Art Laboratory
Our US-based, world-class laboratory contains a robust set of state-of-the-art equipment and analytical instrumentation for every E/L need.


Data Analysis Customized to Meet Your Needs
Our full range of data analytical capabilities covers semi-quantitative, quantitative, and validated methods.
Regulatory Updates
Recently, U.S. Pharmacopeia (USP) approved the revision of two new chapters (665/1665) on production equipment and patient safety for polymeric components and systems used in biomanufacturing. The new USP guidance provides clear expectations for what biomanufacturers using SUS can expect from the regulatory body in their chosen market.
USP <665> is generally aligned with BPOG’s 2020 guidance, including similar or identical component names, categories, and testing time points. The USP chapters are a step forward in finalizing and clarifying regulators’ expectations around fluid-contact equipment in biomanufacturing. The risk-based approach also helps manufacturers prioritize which data they need to present to achieve regulatory success.
With the new USP regulations, regulatory bodies have the prerogative to ask manufacturers to provide whatever data they would like to see. Neither USP nor BPOG possesses enforcement capabilities – their guidance is recommended, not required. On the other hand, the U.S. FDA can enforce its regulations by rejecting device submissions. Biomanufacturers should know that the agency has stated that any primary packaging component that is, or may be, in direct contact with a dosage form needs a safety/toxicological risk assessment. This is a clear indication that the U.S. FDA will likely follow USP’s lead and make chemical characterization, followed by a safety/toxicological risk assessment the standard for SUS and their componentry.