Expert Preclinical and
IND-enabling Testing
Get a comprehensive preclinical testing program that prepares and supports your molecule or new modality through IND-enabling studies and application submission. We provide fully integrated programs that address the bioanalytical, DMPK, and toxicology testing needs of your candidate and meet the demands of your IND submission timeline.
Preclinical and IND-Enabling Testing by WuXi AppTec Lab Testing Division
IND Enabling Packages
In Vitro Studies per Year
In Vivo Studies per Year
Advance Your Candidate Confidently
Through IND Submission
We combine world-class drug development testing with cross-functional program management and global regulatory expertise to shepherd your candidate through IND submission, saving you significant time and expense.
Full-Scale ADME & PK/PD
Leverage a global network of facilities offering a full range of discovery screening, preclinical development, and clinical drug metabolism and pharmacokinetic services.
- Permeability
- Metabolic stability
- Plasma protein binding
- RBC partitioning
- Transporter profiling
- Drug-drug interactions
- Met ID (in vitro/in vivo)
- Mass balance and tissue distribution (QWBA)
- Radiolabeled Synthesis
- PBPK, PK/PD assessment
- Human PK projection
- In vivo efficacy in disease models
- Mechanism of action & translational medicine
Comprehensive Safety Assessment
Our integrated safety assessment services provide multidisciplinary expertise and experience that saves time and cost while yielding the valuable data you need for decision making.
- General toxicology
- Safety pharmacology
- Genetic toxicology
- Developmental and reproductive toxicology (DART)
- Carcinogenicity testing
- Ocular toxicology
- Clinical pathology and anatomic pathology
Pharmacology
Get validated discovery assays for safety pharmacology, including in vitro assays and in vivo disease models in cardiovascular, CNS, respiratory, metabolic, and infectious diseases.
- In vitro assays
- In vivo efficacy in disease models
- Mechanism of action and translational medicine
- Dose-response relationships
Bioanalytical Solutions
Put your small molecule or new modality on the right track with bioanalytical strategy designed to establish and execute the right assays for regulatory submission.
- LC-MS/MS & Immunochemistry platform
- Method development, verification, and validation
- Pre-clinical sample analysis (TK, PK)
- High throughput sample analysis (ADME, in vitro, discovery)
- Biomarker testing
- Immunogenicity assessment
- Customized antibody service
- Clinical bioanalysis
- Central laboratory services
Full CMC & Analytical Development
We support your CMC authoring needs, in both the US and China, from preclinical to commercial lifecycle management for both drug substance and product.
- Process development
- GMP API manufacturing
- Pre-formulation
- Formulation development
- Small scale manufacturing
- Method development & validation
- Compound characterization & batch release
- Stability study
- Regulatory CMC
Regulatory Affairs
Make global filings convenient, efficient, and cost-effective across US FDA, NMPA, and EMA with our extensive regulatory submission support.
- IND feasibility evaluation
- Gap analysis
- Contractual assistance
- Business unit coordination
- Project updates and teleconferences
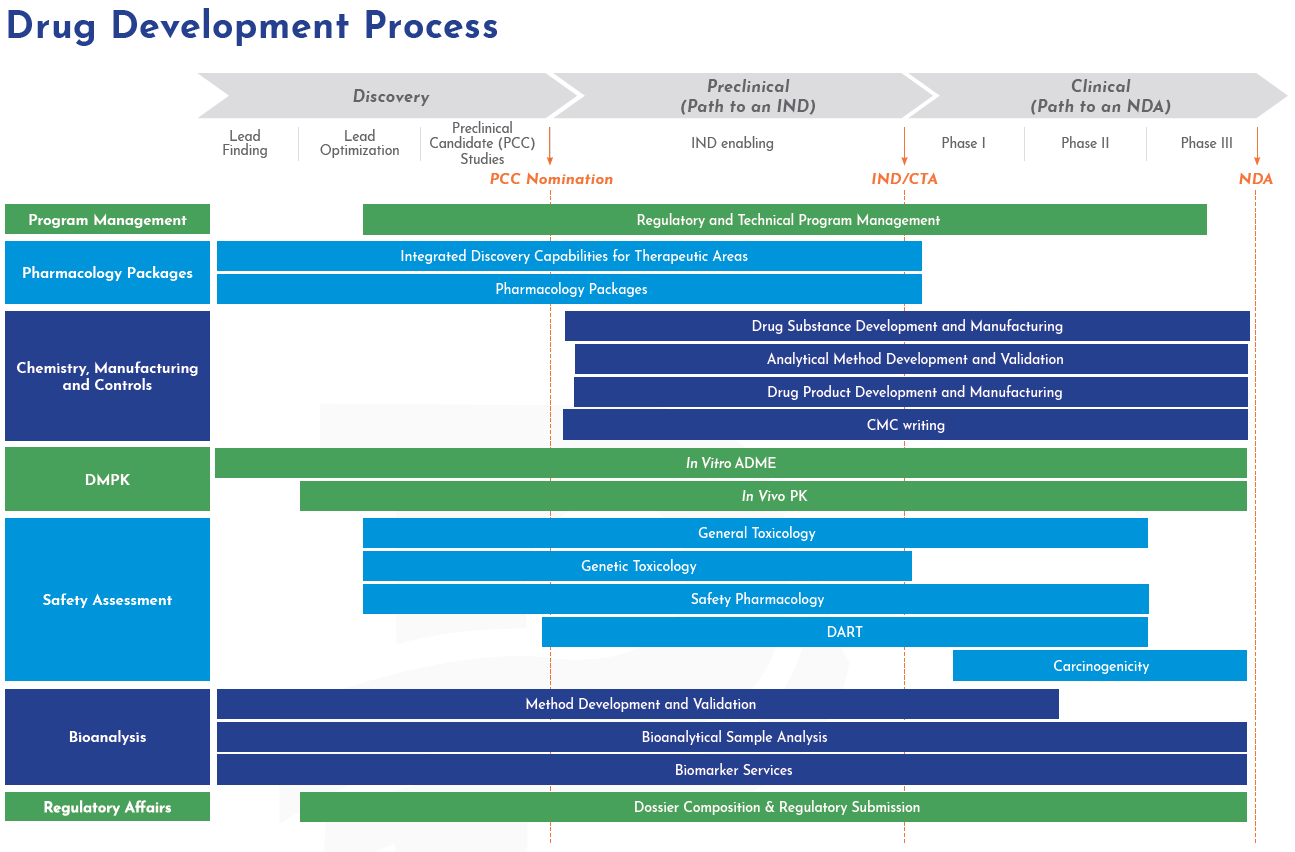
Dedicated IND Program Management
Our program management (PM) team, which includes a dedicated, full-service project manager, supports your IND program end-to-end, including design, timeline determinations, study execution, progress tracking, issue resolution, dossier preparation and regulatory submission.
- Accelerated timeline to expedite your molecule to IND
- Dual filing in U.S. and China to increase asset value
- Truly integrated platform that provides everything you need from one source
- Flexible collaboration that covers you from IND packages to a la carte needs
- High quality service, expertise and facilities that ensure compliance with global regulatory standards

Global Partner & Network
WuXi AppTec is a truly global provider of preclinical small and large molecule solutions, with state-of-the-art facilities and experts across the US, Europe and China. We have decades of experience operating within diverse regulatory environments and delivering tailored testing solutions that advance small and large molecules from preclinical through IND, NDA and beyond.
- 1st CMC platform in China which passed U.S. FDA inspection for new chemical entities
- 1st cGMP biologics manufacturing facility in China compliant with U.S., European and Chinese regulatory standards
- 1st GLP preclinical laboratory in China double certified with an OECD country and NMPA; passed U.S. FDA inspection
- 1st GLP/GCP bioanalytical laboratory in China which passed U.S. FDA, OECD and NMPA inspections
Regulatory Expertise
WuXi AppTec’s regulatory team provides a complete package of services to support global regulatory submission by incorporating internal expertise and partnering with external consultation networks. Our one-stop service can make your global filing convenient, efficient and cost-effective across the U.S. Food and Drug Administration (FDA), National Medical Products Administration (NMPA), European Medicines Agency (EMA), and beyond.
- Regulatory consultation, project feasibility assessment, product registration strategy and planning
- Gap analysis based on available dossier information
- Dossier composition
- Communication meetings with different regulatory agencies
- Electronic Common Technical Document (eCTD) submission
- Coordination of on-site inspection
- National Institute for Food and Drug Control (NIFDC) testing progress follow-ups in China
- Annual reports and subsequent supplement submissions
- Other assistance in product registration
Explore More IND Insights

Frequently Asked Questions
What is IND-enabling testing?
IND-enabling testing refers to preclinical studies conducted to generate data required for the submission of an Investigational New Drug (IND) application to regulatory authorities, demonstrating the safety and efficacy of a drug candidate prior to human clinical trials.
Why is IND-enabling testing necessary for the development of new drugs?
IND-enabling testing is necessary for the development of new drugs because it provides comprehensive preclinical data on a drug candidate’s safety, pharmacokinetics, pharmacodynamics, and toxicological profile of a drug candidate. This data is required by regulatory authorities to assess the drug’s potential risks and benefits before allowing it to proceed to human clinical trials.
What types of studies are typically included in IND-enabling testing?
IND-enabling testing comprises a wide range of preclinical studies aimed at gathering crucial data for regulatory submission. These include full-scale ADME and PK/PD assessments, comprehensive safety evaluations, and pharmacology studies. These efforts ensure the acquisition of robust data essential for informed decision-making and successful regulatory approval.
How does IND-enabling testing differ from other stages of drug development?
IND-enabling testing is a phase of drug development focused on generating preclinical data necessary for an Investigational New Drug (IND) application. It differs from other stages by its primary focus on preclinical assessments and its role in obtaining regulatory approval for human clinical trials.
What are the main objectives of IND-enabling studies?
The main objective of IND-enabling studies is to generate comprehensive preclinical data necessary for submitting an Investigational New Drug (IND) application to regulatory authorities.
What are the regulatory requirements for IND-enabling testing?
Regulatory requirements for IND-enabling testing encompass adherence to Good Laboratory Practices (GLP), compliance with relevant International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines, and the use of scientifically sound study designs and protocols.
How long does IND-enabling testing typically take to complete?
The duration of IND-enabling testing can vary depending on factors such as the complexity of the drug candidate, the scope of testing required, and the specific needs of the client. At WuXi AppTec, we work closely with clients to develop customized IND-enabling testing programs tailored to your specific timelines and requirements.
Can you provide examples of specific tests conducted during IND-enabling studies?
We have extensive experience conducting IND submissions for a diverse range of small molecules and biologics, including antibodies, antibody-drug conjugates, peptides, oligonucleotides, and more. Our track record includes successful completion of over 1,200 IND submissions, showcasing our proficiency in navigating the complexities of preclinical testing and regulatory requirements across various therapeutic modalities.
How does IND-enabling testing contribute to the safety assessment of new drugs?
By generating essential safety data, IND-enabling testing informs regulatory decisions and ensures that only those drug candidates with acceptable safety profiles progress to human clinical trials, thereby protecting patient safety and upholding ethical standards in drug development.
What role does pharmacokinetic and pharmacodynamic (PK/PD) testing play in IND-enabling studies?
PK/PD testing plays a crucial role in IND-enabling studies by providing essential information about how a drug candidate behaves in the body and its pharmacological effects. PK/PD testing data help assess the drug candidate’s absorption, distribution, metabolism, and excretion, as well as its pharmacological effects on the body. These studies help determine optimal dosing regimens, understand drug efficacy and safety profiles, and inform decision-making regarding clinical trial design.
What are the key considerations when selecting a partner for IND-enabling testing?
When selecting a partner for IND-enabling testing, it’s crucial to consider their expertise, experience, flexibility, and track record in conducting preclinical studies. Look for a partner that offers comprehensive services, adheres to quality and compliance standards, and provides customization and flexibility to meet your specific needs. Effective communication, collaboration, and a commitment to innovation are also key factors to ensure a successful partnership in preclinical drug development.
Can IND-enabling testing be conducted in-house, or is it typically outsourced?
IND-enabling testing can be done in-house or outsourced, depending on the organization’s resources and goals. The choice depends on factors like internal capabilities, timelines, and strategic objectives. Both options have pros and cons, so choosing based on what works best for the specific drug development program is important.
What are the potential challenges associated with IND-enabling testing?
Potential challenges associated with IND-enabling testing include the complexity of studies, resource requirements, timeline pressures, regulatory compliance, data interpretation, risk assessment, and interdisciplinary collaboration.
How can IND-enabling testing help mitigate risks associated with clinical trials?
Successfully navigating the risks requires careful planning, effective project management, and collaboration among stakeholders to ensure the timely and accurate completion of preclinical studies and the progression of the drug candidate to clinical trials. Partnering with a reputable service provider for outsourcing testing further enhances efficiency and expertise, offering a strategic solution to mitigate risks and optimize trial outcomes.
Let’s Talk About Your Candidate
Have questions? Need specific assays? Talk to an expert today about your IND-related needs.