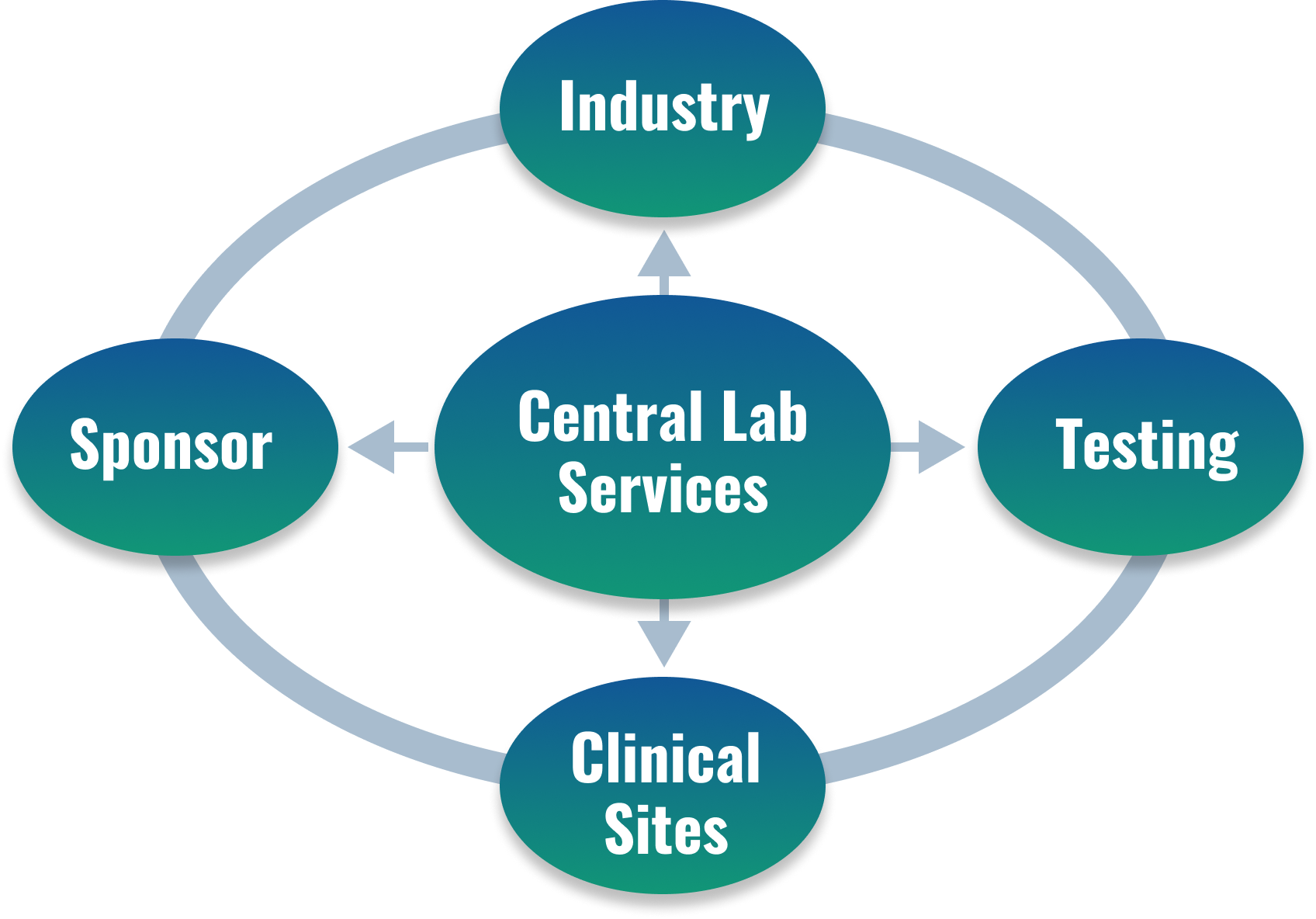
Central Lab Services
WuXi AppTec’s Central Laboratory provides laboratory services that closely connect sponsors, industries, clinical sites and testing labs that support Phase I-IV clinical studies in the global pharmaceutical industry.
Years lab operations in China and the U.S.
Lab space
Samples analyzed per year
Global clients
Global Clinical Trials
Comprehensive Central
Laboratory Services
From analytical method development and validation to sample collection, analysis and data reporting, our experienced professionals collaborate with your team to optimize synergies and efficiencies that produce reliable data and reduce the risk of delays. Our state-of-the-art global facilities are located in Plainsboro, NJ and Shanghai, China.

Dedicated Project Management
Every project is unique and usually involves many stakeholders, so we assign each a dedicated Project Manager (PM). As your primary point of contact, an experienced PM will develop a thorough understanding of your study’s challenges and goals to help deliver tailored and responsive support.
Reliable Sample & Data Management
From sample receipt to testing, sample life-cycle management ensures the integrity of samples. Clinical trials create large volumes of data. Using a world-class laboratory information management system with strict quality control procedures, including a robust backup system, our Central Laboratory ensures the security and integrity of your data.
Adhering to Global Regulatory Standards
We offer high quality services and conform to all the many major global regulations and guidance, including the NMPA, U.S. FDA, OECD, EMA, PMDA and CAP, with an excellent track record of global regulatory inspections.
China NMPA inspections
Internal regulatory inspections
High Quality Standards that We Stand Behind
WuXi AppTec’s Central Laboratory employs highly trained expert personnel and complies with regulations that safeguard the accuracy and reproducibility of your data. Our quality certificates are available upon request.
Robust Clinical Safety Testing
WuXi AppTec’s Central Laboratory can help you monitor the safety of your investigational product by analyzing samples for biological and pathological markers, drug functionality and potential adverse effects. It can also help you determine the participation criteria for patient populations in a clinical study.
Customized Lab Manual
& Kits
Biological sample- and patient-data-integrity are critical. That is why all project personnel train on a manual made specifically for your project that details the materials, protocols, and guidance for proper sample collection, handling, and shipping. Also, each project includes exclusive collection kits with a customized requisition form, pre-labeled vials and collection and storage supplies.
-
Custom-designed for each study
-
Specific to site and visits
-
Pre-populated with site address
-
Site personnel simply completes the patient information in the prompts provided

Global Logistics Capabilities
When conducting country-specific studies, enjoy an industry-leading turnaround time and have confidence in the integrity of your samples with WuXi AppTec’s global logistics program. Our consolidated network with standardized services features 24 hours or less delivery within North America and 48 hours or less globally with a record of greater than 98% on-time delivery worldwide.
Frequently Asked Questions
What are Central Laboratory services?
In clinical trials for new drugs and other therapeutic interventions, a Central Laboratory is the operational hub. It connects the sponsor, industry, clinical sites and testing labs. It provides the necessary materials, protocols, and guidance for proper sample collection, handling, and shipping that safeguard the integrity of all biological samples. A Central Laboratory is also responsible for sample testing and analyzing, reporting, and archiving the data generated, which is vital to the regulatory approval process.
What is the benefit of using Central Laboratory services?
A Central Laboratory eliminates the need to work with multiple vendors, giving you more time to focus on other priorities. Also, bundled services and access to expert resources, plus the synergies and efficiencies that result from working with a single point of contact, often result in financial savings.
When do I need Central Laboratory services?
A Central Laboratory should be engaged before initiating your Phase I-IV clinical studies. While individual services may be an option, the complexity of most clinical trials typically necessitates a comprehensive partnership. For example, it’s a considerable challenge to coordinate the collection, tracking and storage of potentially hundreds – even thousands – of samples from many different sites from many countries, let alone processing, analyzing, reporting, and archiving the results.
How do Central Laboratory services aid regulatory submission?
A Central Laboratory usually helps a client collect and analyze samples and to report and archive the data used in a submission for an Investigational New Drug (IND), Clinical Trial Application (CTA), New Drug Application (NDA) and/or Biologics License Application (BLA). Project personnel may also guide study design, execution and reporting.
What services are typically included in Central Laboratory services?
Material and Sample Management
- Kit material management
- Sample collection kits assembly
- Sample management
- Sample storage
Project Management
- Lab manual
- Site training
- Logistic and global shipping management
- Country specific import & export support
- Regional regulatory support for Customs clearance
Lab Testing
- Standardized Diagnostic/Safety/Specialized testing
- Fast turn-around and reliable data and reports
- Regulatory inspection support
Need a Central Lab Partner? Let’s Talk
Find out how our Central Laboratory Services can help your clinical studies.