PROteolysis TArgeting Chimera, the novel therapeutic modalities better known as PROTACs*, first emerged in 2001 but are just starting to emerge from clinical trials. Their unique structure and capabilities have excited drug developers; however, there are still questions about their broad-scale utility and many challenges to overcome when tackling DMPK (drug metabolism and pharmacokinetics) studies.
Like the mythical three-headed beast they’re named after, a PROTAC consists of three structural parts. The first is a ligand that binds to the ubiquitin-protein ligase E3. The second is a ligand that binds to a target protein earmarked for degradation. The final part is a covalent linker connecting two ligands.
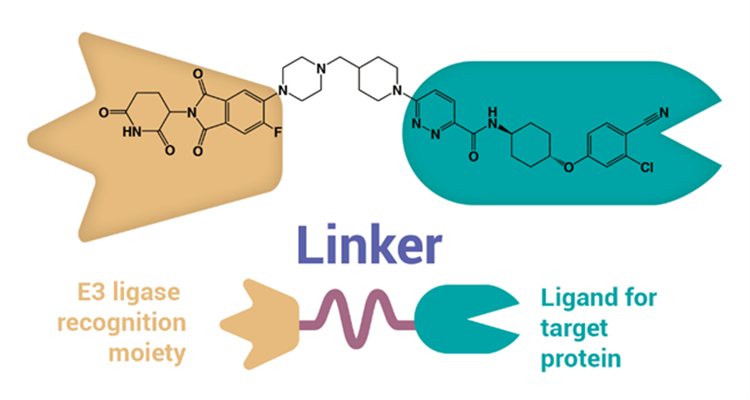
How do PROTAC Drugs Work?
Put very simply, PROTACs use the cell’s own waste disposal system to attack and treat disease. They don’t delete target proteins themselves but instead use the natural cell protein degradation system to break them down.
The unique mode of action differentiates them from conventional small molecules and allows them to access previously inaccessible proteins and treat diseases once thought untreatable. Their ability to selectively degrade pathological proteins gives them a massive advantage over other small molecules, spurring drug developers into action.
DMPK Challenges in PROTAC Drug Development
Despite the excitement over the potential for the future of PROTAC drugs, development still comes with significant challenges. The list of DMPK hurdles developers must overcome is lengthy.
- Regulatory agencies are not supplying any guidance on how to address DMPK issues. Guidance for molecule drugs is usually taken on a case-by-case basis, but PROTACs are different from small molecule drugs.
- PROTACs’ unique structure makes it challenging to meet Lipinski’s Rule of Five, which is designed for small molecules. PROTAC drugs have poor permeability, which means poor druggability for oral administration. And the correlation between in vitro and in vivo permeability is weak.
- PROTAC drugs have shown a “hook effect,” so the drug research community must understand PK/PD characteristics for PROTAC drugs and the proper dose ranges more clearly.
- PROTAC’s metabolites may compete for binding targets or E3 ligases, reducing efficacy. That makes metabolite profiling and identification a crucial part of preclinical screening.
Strategies for PROTAC DMPK Studies
Because of the challenges associated with PROTACs, drug developers should ensure consistent optimization of physicochemical and DMPK properties when designing a preclinical evaluation strategy. This needs to be performed during the early screening stage by first characterizing the in vitro properties of PROTAC molecules.
In the optimization stage, the focus should be on improving the metabolic clearance and solubility of PROTAC, combined with the PK properties of extravascular drug delivery, to understand better the DMPK properties of PROTAC, such as absorption and metabolism. The permeability of PROTACs is a challenge for improving due to their structure. Orally administered PROTACs will require more attention to solubility and metabolic stability during this phase.
At the PCC stage, PROTAC molecules with potent and better oral bioavailability can be used in further PK/PD studies to obtain a more in-depth exposure-response relationship.

Cause for Optimism
As investigations into PROTAC evolve, scientists will discover better techniques to mitigate some of the challenges drug developers face today. There are also some strategies that developers can adopt to help speed up these efforts:
- Screen compounds with caution
- Balance its hydrophilic and lipophilic
- Try to be close to Lipinski’s Rule of Five
- Chose E3 ligases carefully
- Seek out new protein targets
The Bottom Line
PROTAC drugs have generated a lot of excitement, and understandably so. Their unique mechanism— and rapid progress through clinical trials—is evidence that they hold immense potential for treating some of the worst diseases. But there are challenges to overcome, particularly in the field of DMPK. If these hurdles can be overcome, the vast potential of PROTACs will be unlocked. Open communication and collaboration between laboratory testing partners, academic institutions and drug developers and sponsors can push the field forward and deliver these therapeutic options to those who need them most.
Reference:
[1] Cantrill C , Chaturvedi P , Rynn C , et al. Fundamental aspects of DMPK optimization of targeted protein degraders. Drug Discovery Today, 2020, 25(6):969-982.
*PROTAC® is a registered trademark of Arvinas. In this article, PROTAC specifically refers to the abbreviation of PROteolysis TArgeting Chimera as therapeutic modalities.
As a global company with operations across Asia, Europe, and North America, WuXi AppTec provides a broad portfolio of R&D and manufacturing services that enable the global pharmaceutical and life sciences industry to advance discoveries and deliver groundbreaking treatments to patients. Through its unique business models, WuXi AppTec’s integrated, end-to-end services include chemistry drug CRDMO (Contract Research, Development and Manufacturing Organization), biology discovery, preclinical testing and clinical research services, helping customers improve the productivity of advancing healthcare products through cost-effective and efficient solutions. WuXi AppTec received an AA ESG rating from MSCI for the fourth consecutive year in 2024 and its open-access platform is enabling around 6,000 customers from over 30 countries to improve the health of those in need – and to realize the vision that “every drug can be made and every disease can be treated.”