Genetic Toxicology
Genotoxicity is integral to the toxicological information package used in drug development decision-making and risk assessment. WuXi AppTec’s Genetic and In Vitro Toxicology (GIVT) department offers diverse genotoxicity assays—from early screening to IND filing—and is a one-stop shop for genotoxicity evaluation.
Genetic Toxicology Services by WuXi AppTec Lab Testing Division
Years of Genetic Toxicology Experience
Safety Assessment Staff
Safety Assessment Studies Conducted/Year
Genetic Toxicology Testing & Evaluation
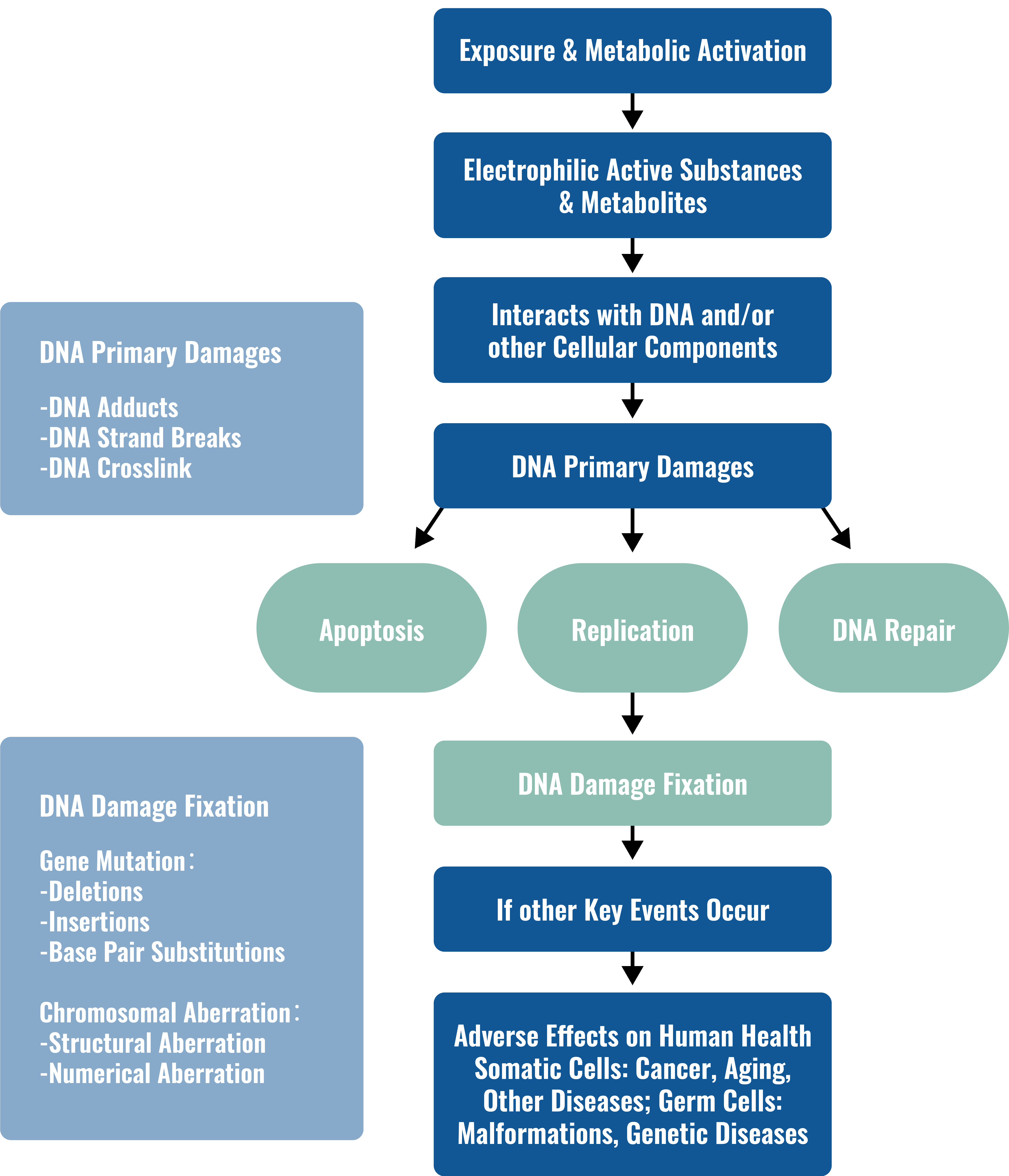
When DNA is exposed to particular test articles, mutations and other damage can occur, leading to cancer and/or teratogenic effects. The severity of these effects necessitates examining whether new or existing test articles intended for human use have any effect on DNA. No single test can detect all relevant genotoxic endpoints, so regulatory agencies recommend a battery of in vitro and in vivo tests for genotoxicity.
From Exposure to Genotoxic Responses
Genetic Toxicology Testing Capabilities
Founded in 2009, WuXi AppTec’s state-of-the-art laboratory serves worldwide customers with genotoxicity. The laboratory passed the GLP inspections by FDA, OECD and NMPA. GLP and non-GLP studies are conducted in compliance with ICH, OECD and NMPA guidelines.
Genetic Toxicology Screening Battery
A standard genetic toxicology screening battery includes screening bacterial reverse mutation assays (Ames), and screening in vitro micronucleus assays. WuXi AppTec offers (Q)SAR mutagenicity prediction for drug discovery and impurities as well.
Bacterial Reverse Mutation Assays (Ames)
Bacterial reverse mutation assays (commonly known as Ames) are genetic toxicology assays that identify substances that induce gene mutations. WuXi AppTec offers two formats for Ames assays:
Agar format
- Micro-Ames assay, 24-well plate
- Mini-Ames assay, 6-well plate
- Ames assay, petri dish
Liquid format
- Ames II
- Ames microplate format (MPF)
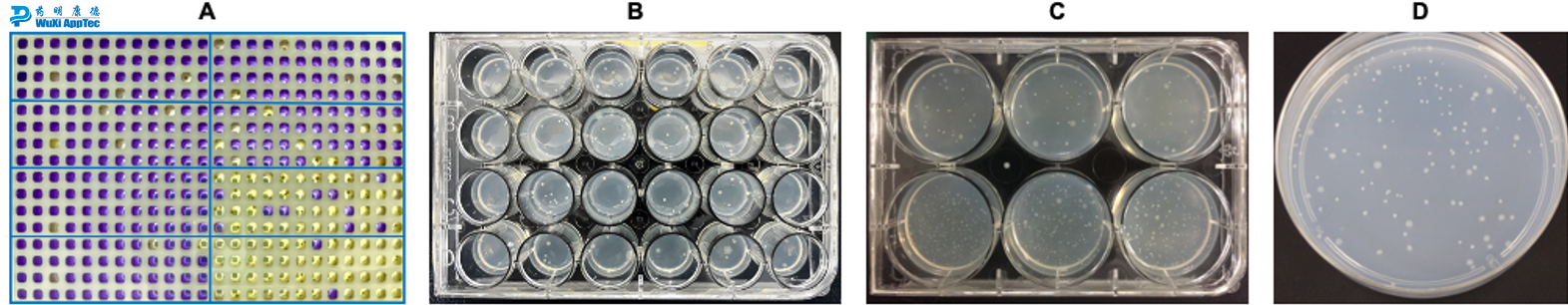
Liquid format Ames II (A); Micro-Ames in 24-well plate (B); Mini-Ames in 6-well plate (C); Standard Ames in Petri Dish (D).
Screening Micronucleus Assay
The in vitro micronucleus assay identifies substances that induce clastogenicity or aneugenicity. Clastogenicty is a structural chromosomal aberration through breaks in DNA. Aneugenicity is a numerical chromosomal aberration through interactions with non-DNA cellular targets. WuXi AppTec deploys the following methods:
- CHO Microwell MNT, manual method
- MicroFlow, flow cytometer method
(Q)SAR Mutagenicity Prediction
The International Conference on Harmonization (ICH) M7 guidelines for assessing and controlling DNA-reactive (mutagenic) impurities in pharmaceuticals allow for the consideration of in silico predictions in place of in vitro studies. This represents a significant advance in the acceptance of Quantitative Structure-active Relationship (Q)SAR models.
The QSAR mutagenicity prediction is an in silico tool for predicting the mutagenicity of compounds, impurities, and metabolites. Mutagenicity is the potential for a test article to cause genetic mutations. For impurity evaluation and drug discovery, WuXi AppTec uses:
- An expert rule-based model, Leadscope Model Applier
- A statistical model, Leadscope Model Applier
Genetic Toxicology GLP Battery
IND-enabling testing routinely includes a series of toxicology studies, including genetic toxicology. A standard GLP battery for genetic toxicology includes an Ames assay, in vitro mammalian cell assay, in vivo genotox assay, and mechanism exploration and follow-up assays.
Ames Assay
Ames (bacterial reverse mutation assays) identifies substances that induce gene mutations. WuXi AppTec’s GLP Ames methods include:
- Plate incorporation
- Pre-incubation
- Treat and wash to avoid feeding effect
- Enhanced Ames for N-nitrosamines
In vitro mammalian cell assay
Mammalian cell assays evaluate whether a chemical substance has the potential to cause genetic damage to mammalian cells. These assays include:
In vitro chromosomal aberration
In vitro micronucleus (TK6 or CHO)
Mouse lymphoma assay (MLA)
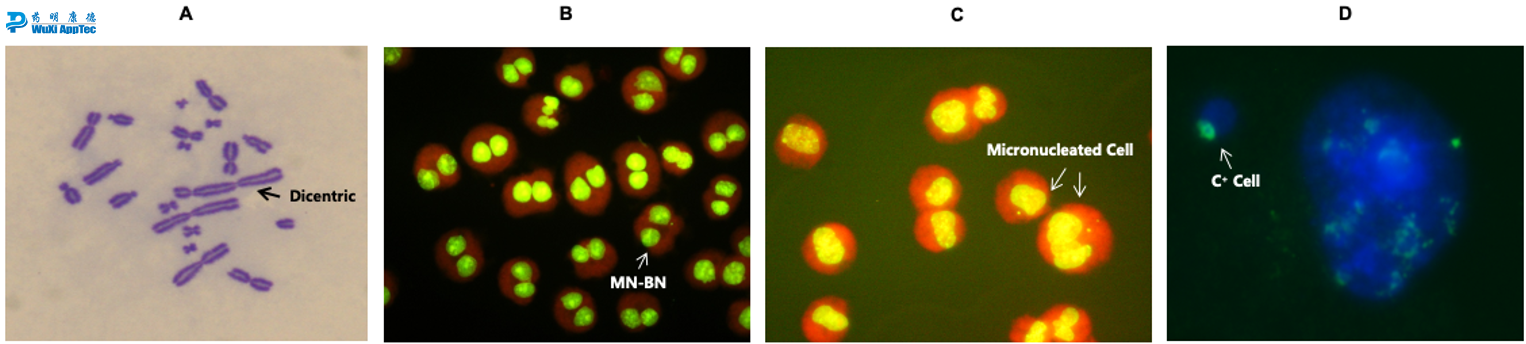
Images from in vitro cytogenetic assays. (A) Chromosomal damage observed in metaphase spreads using CHO-WBL cells; (B) Examples of binucleated cells with micronuclei (MN-BN) in the microwell micronucleus assay with CYB; (C) Fluorescent microscope images of micronucleated cells in the micronucleus assay without cytochrome B (CYB); (D) Micrograph of FISH analysis with pancentromeric probes applied to the micronucleus assay.
In vivo genotox assay
In vivo genotoxicity assays evaluate the potential of substances to cause genetic damage.
- In vivo micronucleus (manually or FCM based method, standalone or integrated)
- In vivo Comet, option 2
Mechanism exploration & follow-up assays
WuXi AppTec offers a series of additional genetic toxicology assays that complement the standard battery of GLP in vitro and in vivo assays, providing additional insights into how test articles might impact human DNA.
- In vivo chromosomal aberration
- In vivo Comet (standalone or integrated)
- In vivo Pig-a
- Fluorescence in situ hybridization (FISH)
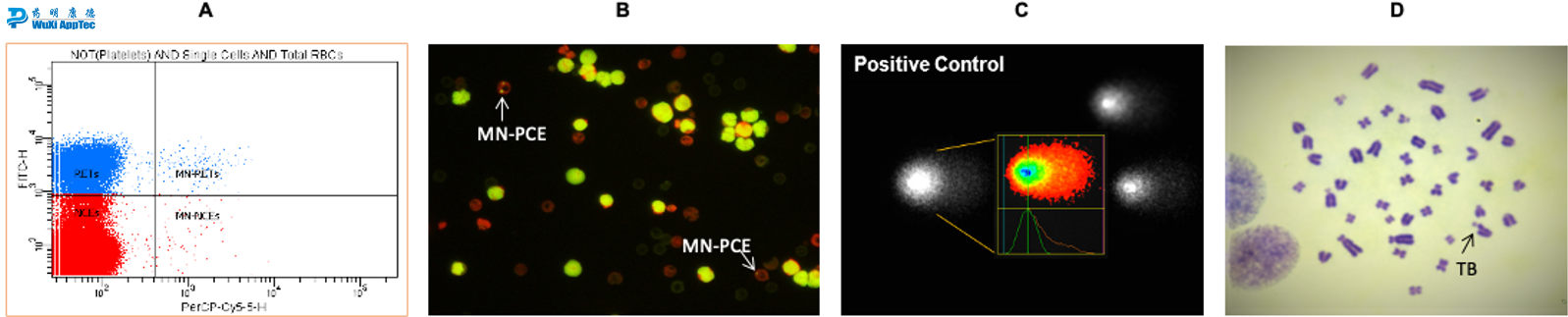
Images from in vivo genetic toxicology assays. (A) Automated analysis of the formation of micronuclei in reticulocytes analyzed by flow cytometry; (B) Fluorescent microscope images of micronucleated PCEs; (C) Images of Comet analyzed by Cyto Study Manager System; (D) Chromosomal damage observed in metaphase spreads
Genetic Toxicology Reporting
WuXi AppTec leverages an automated reporting system to draft reports and has a dedicated study director group for report checking and peer-reviewing. Our team is committed to provide high quality study data and deliver the reports timely in order to meet your timeline.
Frequently Asked Questions
What is In Vitro Toxicology Testing?
It’s crucial for identifying potential genotoxic risks, which can lead to serious health issues such as cancer and genetic diseases. Ensuring the safety of substances before they are widely used protects public health and meets regulatory requirements.
Why is genetic toxicology testing important?
It’s crucial for identifying potential genotoxic risks, which can lead to serious health issues such as cancer and genetic diseases. Ensuring the safety of substances before they are widely used protects public health and meets regulatory requirements.
What are the primary methods used in genetic toxicology testing?
Key methods include the Ames test, chromosomal aberration test, and micronucleus test. Each method evaluates different aspects of genotoxicity to provide a comprehensive safety profile.
What is the Ames test?
The Ames test detects mutations in bacteria caused by chemical substances, indicating potential genotoxicity. It is a widely used and accepted method for initial genotoxicity screening.
What is the micronucleus test?
This test measures chromosomal damage by counting micronuclei in cultured cells/rodents, which indicate genotoxic effects. It is valuable for assessing both clastogenic and aneugenic effects of substances.
What is the chromosomal aberration test?
This test examines cells for structural changes in chromosomes, which can indicate genotoxicity. It helps identify substances that may cause cancer or heritable genetic defects.
When should genetic toxicology testing be conducted?
It’s typically conducted during early drug development and preclinical stages. Identifying potential genotoxic risks early helps avoid costly failures in later stages of development.
What is a GLP battery in genetic toxicology testing?
A GLP (Good Laboratory Practice) battery includes a series of standardized tests conducted under strict regulatory guidelines. These guidelines ensure the reliability, reproducibility, and integrity of test results.
How does genetic toxicology testing contribute to IND filing?
It provides essential safety data required for IND filing, demonstrating that the drug is unlikely to cause genetic harm in humans. This data is critical for gaining regulatory approval to proceed with clinical trials.
What is the risk of genetic toxicity?
Genetic toxicity can lead to mutations, cancer, and heritable genetic changes, posing serious health risks. Understanding and mitigating these risks is essential for developing safe pharmaceuticals and chemicals.
How can early screening in genetic toxicology testing benefit drug development?
Early screening helps identify genotoxic risks early in the development process. This allows for modifications to the substance or development strategy, saving time and resources.
What regulatory guidelines govern genetic toxicology testing?
Regulatory guidelines are set by agencies like the FDA, EMA, and ICH. These guidelines ensure standardized and reliable testing methods that protect public health.
How can I choose the right genetic toxicology tests for my substance?
Consulting with experts and considering the substance’s nature, intended use, and regulatory requirements can help select appropriate tests. Tailoring the testing strategy to the specific needs of the substance ensures comprehensive safety assessment.