Repeat Dose Toxicity Testing
Repeat dose or chronic toxicity studies help evaluate the effects of repeat administration of a new drug candidate over a defined period of time. Chronic toxicity studies help you select proper dosing amounts for GLP studies and identify potential target organs affected by the compound. As an experienced partner in general toxicology testing, WuXi AppTec can help you build a safety assessment program that includes the correct repeat dose toxicity studies for your needs.
General Toxicology Studies Performed
Square Feet in Facilities
almost 500
Study Rooms
Repeat Dose Toxicity Testing Capabilities
WuXi AppTec offers end-to-end toxicology testing for all stages of the drug development lifecycle, including IND-enabling, subchronic, and chronic repeat dose studies.
Repeat Dose Toxicity Study Species & Routes of Administration
Our experienced team can help you design repeat dose toxicity studies with the right toxicological species, dose level, routes of administration, analyses, and regulatory support.
Species
- Canine
- Mouse
- Rat
- Rabbit
- NHP
- Swine
Routes of Administration
Our cutting-edge facilities are stocked with some of the most advanced bioanalytical equipment in the world.
- Intravenous (bolus, infusion)
- Oral (gavage, capsule) and nasogastric (for NHPs)
- Topical
- Intramuscular
- Subcutaneous
- Intradermal
- Ocular (including systemic, topical, subconjunctival, intravitreal, sub-retinal and retrobulbar)
- Implant
- Other (upon request)
- Intrathecal
- Intra-articular
- Intraperitoneal
- Intra-arterial
Other General Toxicology Studies
Repeat dose chronic toxicity studies are part of a complete general toxicology program. Evaluate the safety and potential adverse effects of new compounds and products with WuXi AppTec’s comprehensive service platform.
Repeat Dose Toxicity Data
Repeat dose toxicity studies generate data that deepen researchers’ understanding of dose and toxicity for a given compound. This chronic toxicity data helps identify correct dosing for the pivotal later stage GLP studies. Repeat dose toxicity studies are a critical part of the drug development lifecycle, playing important roles through the discovery, preclinical to IND, and clinical stages.
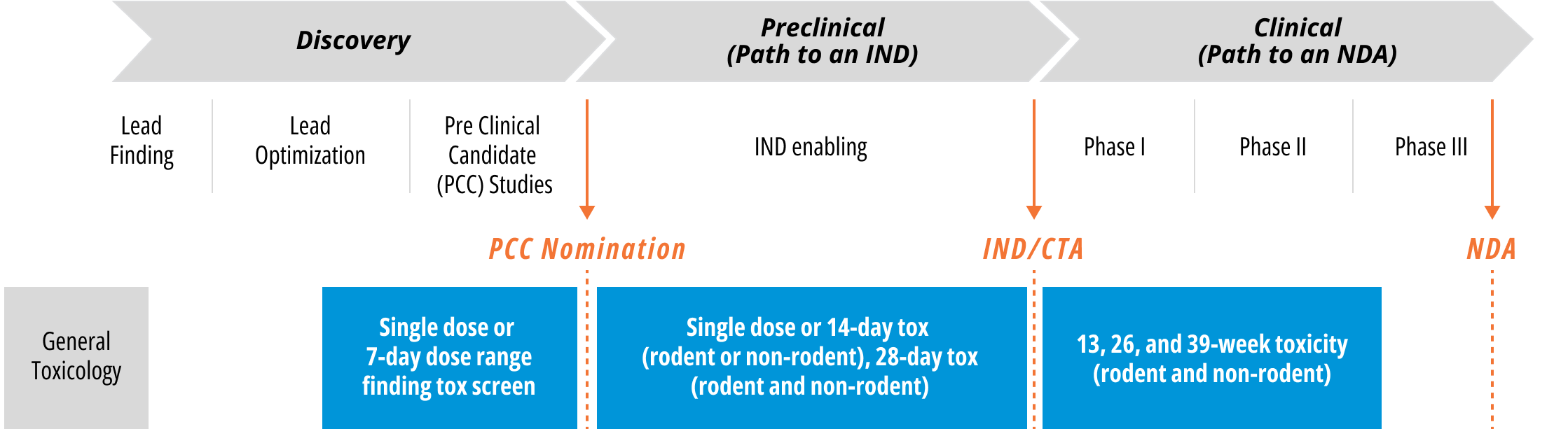
Repeat Dose Toxicity Study Durations
The U.S. FDA’s “M3(R2) Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals” provides guidance on the recommended length of repeat dose toxicity studies. WuXi AppTec can help you select the right duration that matches or exceeds your clinical dosing period.
Acute
Determines if subjects can tolerate a dose level over a shorter time period.
Timeframe
- 2-weeks or less
Sub-Chronic
Shows a more complete picture, including hematology and histology.
Purpose
- Clinical chemistry
- Hematology affects
- Clinical observations
- Histology
Timeframe
- 4 to 13 weeks
Chronic
Assesses the potential for lifelong health impacts from repeat exposure.
Purpose
- Chronic diseases
- Carcinogenicity
Timeframe
- 6 to 9 months
- 1-2 years+
Explore Related Resources About Repeat Dose Toxicity & General Toxicology Testing
Repeat Dose Toxicity Studies FAQ
What are repeat dose toxicity studies?
Repeat dose toxicity studies test the safety of a new drug candidate by exposing animals to repeated doses of the compound and monitoring for signs of toxicity over the course of 14-28 days.
Why are repeat dose toxicity studies necessary?
Repeat dose toxicity studies evaluate the effects of repeat administration over a defined period of time. The studies will provide detailed information about the potential target organs of toxicity, the exposure, as well as the potential of toxic effects.
How do repeat dose toxicity studies add value to my testing program?
The studies will provide information about the toxicity of the organs effected by the compound. The repeated-dose toxicity studies are essential for the quantitative risk assessment of drug molecules.



