Gene Therapy
Speak to an Expert
about how we can help with your upcoming project
End-to-End Gene Therapy Services
From DNA to IND and beyond, WuXi AppTec offers fully comprehensive, end-to-end services for AAV-based gene therapy products.
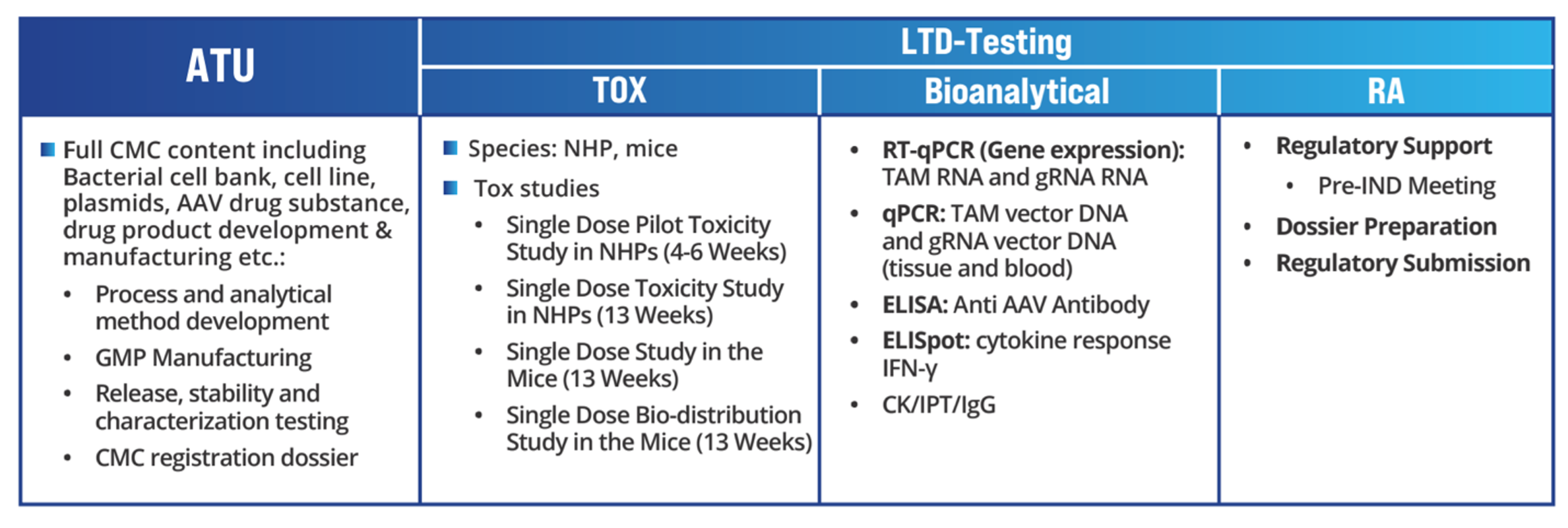
Toxicology
WuXi AppTec’s Saftey Assessment gene therapy services are fully compliant with the requirements of the FDA, OECD, and NMPA GLP. Studies are conducted in state-of-the-art facilities with 500+ animal rooms that are fully AAALAC-accredited and overseen by highly experienced Testing Facility Management experts.
Studies
- General toxicology
- Specialty toxicology
- Local toxicity
- Safety pharmacology
Standard Routes of Administration
- Intravenous (IV)
Non-standard Routes of Administration
- Intrathecal
- Intra-cisterna magna
- Sub-retinal
- Intraarticular
Bioanalytical Services for Gene Therapy
WuXi AppTec’s Bioanalytical group fully covers the needs of gene therapy development.
Pharmacology
- Neutralizing Antibody Prescreening (MSD, ELISA)
- Target Protein Detection (MSD, ELISA, LC-MS/MS)
- Enzyme Activity of Target Protein (Microplate reader, LC-MS/MS)
Pharmacokinetics
- Delivery Vector (LNP) (LC-MS/MS)
- Nucleic Acid (LC-MS/MS, qPCR, ddPCR, Hybrid-ELISA)
- Biodistribution (LC-MS/MS, qPCR, ddPCR, Hybrid-ELISA)
Toxicology & Toxicokinetics
- Immunogenicity (MSD, ELISA)
- Immunotoxicity (FACS, MSD, ELISA, Luminex, ELISPOT)
- Shedding (ddPCR)
Gene Therapy Development Timeline
WuXi AppTec tailors each gene therapy development program to the needs of the material and regulatory guidelines. By utilizing the single platform and internal expertise of WuXi AppTec, the overall timeline can be reduced.
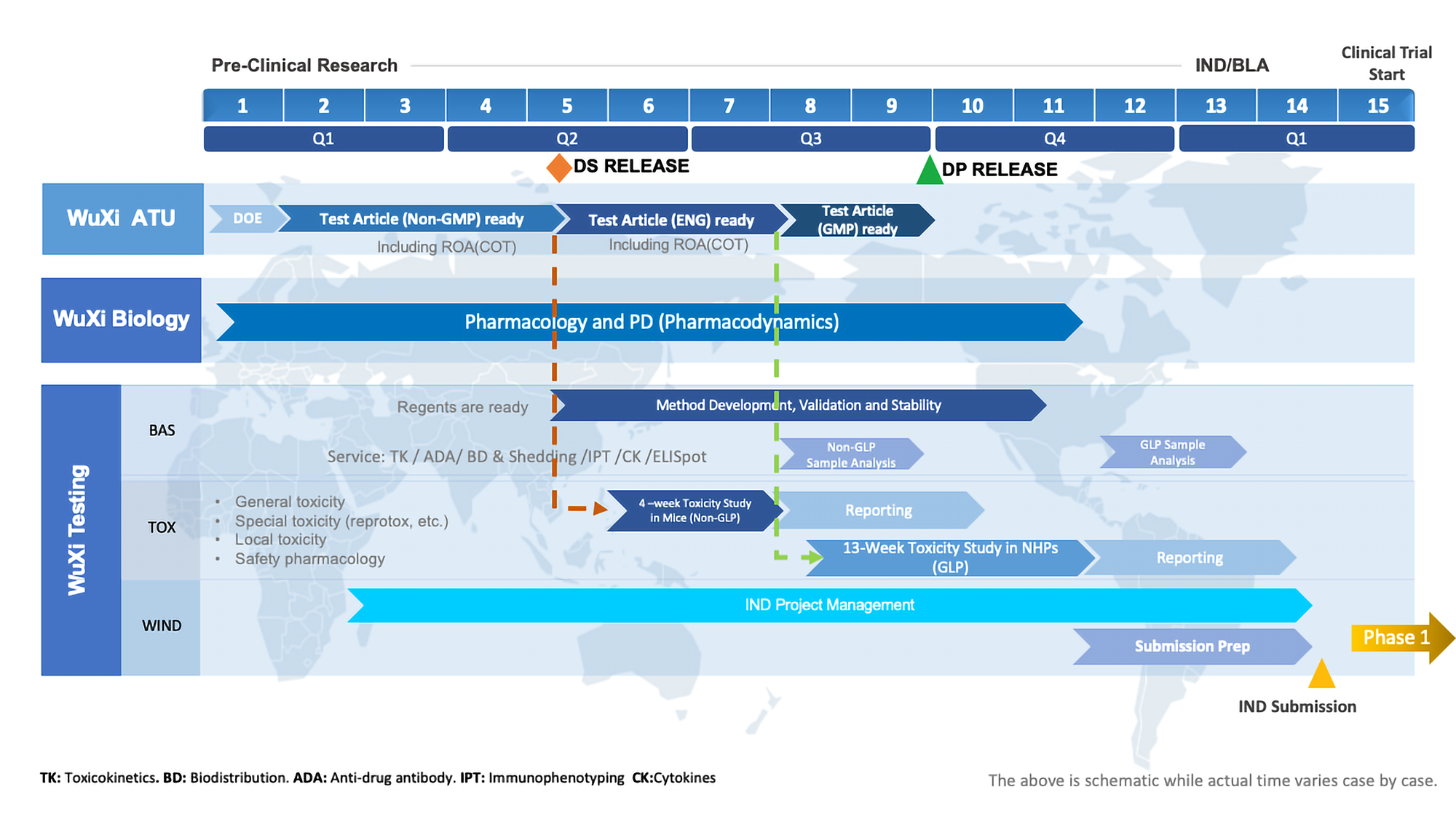
Gene Therapy Case Study
AAV-Based
- Indication: Rare disease with single injection
- Submission plan: FDA & NMPA