Analytical Services
Full Range of Analytical & Regulatory CMC Services
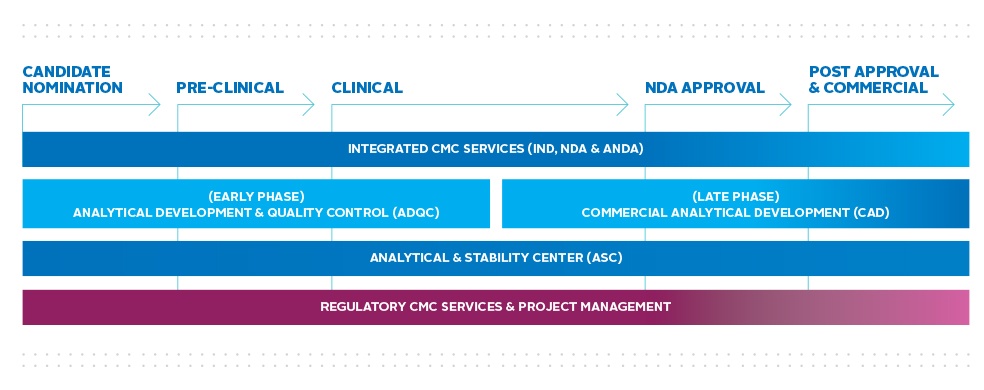
The Analytical Service Unit supports clients from preclinical phases through market supply. Our flexible analytical solutions are available as part of an IND- or NDA-enabling package, as well as on a standalone basis. As part of our integrated approach, the Analytical Service Unit works in close collaboration with the DMPK, Bioanalytical and Toxicology units within the Laboratory Testing Division, allowing our highly-skilled teams to leverage their complementary capabilities to deliver the highest quality data with speed and efficiency.
Service & Quality
Our analytical programs include API and drug product method development and validation and release, manufacturing support, IND and NDA enabling stability studies, reference standard qualification, impurity control, preformulation and product characterization. Our technical writers provide regulatory CMC support by composing IND and NDA packages, and have a proven record of successful submission of these packages to global regulatory agencies. Our commitment to world-class quality has been validated by government agencies as well as numerous clients, and we adhere to strict project and process management standards for all studies.
- Early & Late Phase Analytical Development
- Stability Studies
- Impurity Research
- Highly Potent Compound Analysis
- Large Molecule Analysis
- Microbial Testing
- Preformulation Study
- Physiochemical Characterization & Excipient Release
- Regulatory CMC

Dedicated Facility
Based in Shanghai, China, the Analytical Service Unit has 8,500 square meters of laboratory space, with a capacity of approximately 1,000 cubic meters of available chamber storage. The facility houses state-of-the-art instruments to support analytical development and stability studies for both small and large molecule products.
Track Record of Success
From method development and validation to NDA preparation and submission, the Analytical Service Unit is your single solution for analytical development and regulatory support, having conducted stability programs for more than 2,000 batches of API and drug product and preparing over 50 CMC dossiers to date.
Customers for Discovery Chemistry & Separation, API & Clinical Trial Material Manufacturing
INDs / NDAs Submitted to FDA
Stability Programs Conducted for 1,000 Batches of API & Drug Products
CTDs / CTAs Submitted to CFDA
Operations & Compliance
The Analytical Services Unit operates in full compliance with FDA, MPA, CFDA and GMP requirements, which is validated through regular inspections by both regulatory authorities as well as our clients. Each of our employees undergoes a systematic training regimen that covers analytical sciences, GMP compliance, workflow operations and standard operating procedures to ensure our quality standards are consistently being met across the division.
- 220+ GMP Client-Initiated Audits Since 2009
- FDA Inspections in 2014 and 2016
- MPA Inspections in 2009, 2012 and 2016
- Numerous CFDA Inspections
- Electronic Data Handling Compliant with FDA 21 CFR Part 11
- Periodic Inspections & Data Completeness Review from Independent QAU
- Highly Capable Team of 400+ Staff Across Analytical Development, Stability and Regulatory CMC
